Think It Over (Page No. 162)
1. Water can be obtained from various sources. Are all these samples of water chemically identical?
Answer: Yes, all samples of purified water are chemically identical, regardless of their source — whether from rivers, borewells, rainwater, or the ocean. This is explained by the Law of Constant Proportions (Proust’s Law), which states that a compound always contains the same elements combined in a fixed ratio by mass. Water always contains hydrogen and oxygen in a mass ratio of 1:8, no matter where it comes from. So if 9 g of purified water is decomposed, it always gives 1 g of hydrogen and 8 g of oxygen.
2. Oxygen is sometimes represented as O and sometimes as O₂. What is the difference between these symbols?
Answer:
- O represents a single oxygen atom — it is just one atom of the element oxygen. It is an isolated atom and does not exist independently under normal conditions.
- O₂ represents an oxygen molecule — it consists of two oxygen atoms joined together by a double covalent bond. Each oxygen atom has 6 valence electrons and needs 2 more to complete its octet, so two oxygen atoms share two electrons each to form a stable molecule. O₂ is the form in which oxygen naturally exists in the atmosphere.
In short, O is an atom, while O₂ is a molecule.
3. Why does dissolved salt in water conduct electricity, but sugar does not?
Answer:
- Salt (Sodium Chloride — NaCl) is an ionic compound. When dissolved in water, it dissociates into free-moving ions — sodium ions (Na⁺) and chloride ions (Cl⁻). These freely moving ions carry electric charge through the solution, allowing it to conduct electricity.
- Sugar is a covalent compound. When dissolved in water, it does not break into ions — it simply dissolves as neutral molecules. Since there are no free ions to carry charge, the sugar solution does not conduct electricity.
In summary, electrical conductivity in a solution depends on the presence of free ions, which ionic compounds provide but covalent compounds like sugar do not.
Pause and Ponder (Page No. 166)
1. A student burns 10 g of ethanol in an open beaker. After the reaction, no residue is left in the beaker. Does this mean the Law of Conservation of Mass is violated? Explain.
Answer: No, the Law of Conservation of Mass is NOT violated.
When ethanol burns, it reacts with oxygen from the air and produces carbon dioxide and water vapour. Since both products are gases, they escape into the atmosphere and are not visible in the beaker, which is why no residue remains.
The Law of Conservation of Mass states that mass can neither be created nor destroyed in a chemical reaction. The mass has not disappeared — it simply escaped as gases. If the experiment were performed in a closed system, the total mass of products would equal the total mass of reactants, proving the law is obeyed.
2. When 20 g of hydrogen reacts completely with 160 g of oxygen, how much water is formed according to the Law of Conservation of Mass?
Solution:
According to the Law of Conservation of Mass:
Mass of Reactants = Mass of Products
- Mass of hydrogen = 20 g
- Mass of oxygen = 160 g
- Total mass of reactants = 20 + 160 = 180 g
Hydrogen + Oxygen → Water
Therefore, mass of water formed = 180 g
Pause and Ponder (Page No. 167)
3. A compound consists of 40% sulfur and 60% oxygen by mass. In a sample of the same compound containing 20 g of sulfur, what mass of oxygen must be present to satisfy the Law of Constant Proportions?
Solution:
Given:
Sulfur = 40%
Oxygen = 60%
Ratio of sulfur : oxygen = 40 : 60 = 2 : 3
If sulfur = 20 g, then oxygen =
30 g of oxygen is required.
4. Carbon monoxide (CO) contains carbon and oxygen in the mass ratio of 3:4. How much oxygen will combine with 9 g of carbon to form carbon monoxide?
Solution:
Given ratio of carbon : oxygen = 3 : 4
If carbon = 9 g, then oxygen =
12 g of oxygen will combine.
5. The Law of Definite Proportions holds true for compounds but not for mixtures. Give reason.
Answer: The Law of Definite Proportions states that a compound always contains the same elements combined in a fixed ratio by mass, regardless of its source or method of preparation.
This is true for compounds because they are formed by chemical combination of elements in a definite and fixed ratio. For example, water always contains hydrogen and oxygen in the ratio 1:8 by mass.
However, mixtures do not follow this law because their components can be combined in any ratio. For example, you can mix sand and water in any proportion — there is no fixed ratio. Since no chemical bonding occurs in mixtures, the composition can vary freely.
6. Students X and Y, both prepared an oxide of copper by combining copper and oxygen in the ratios of 4:1 and 8:2, respectively. Do their results justify the Law of Constant Proportions? Explain.
Answer: Yes, their results justify the Law of Constant Proportions.
Student X combined copper and oxygen in the ratio 4:1
Student Y combined copper and oxygen in the ratio 8:2
Simplifying the ratio of Student Y:
8:2 = 4:1
Both ratios are the same when simplified. This means that both students obtained the same compound with the same fixed ratio of copper to oxygen by mass, regardless of the quantities used.
This confirms the Law of Constant Proportions, which states that a compound always contains its elements in a fixed ratio by mass, no matter how it is prepared or in what quantity.
Pause and Ponder (Page No. 168)
7. Assertion (A): 2 g of hydrogen combines with 16 g of oxygen to form 18 g of water.
Reason (R): According to Dalton’s Atomic Theory, atoms combine in a simple whole number ratio by mass to form compounds.
Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Answer: (ii) Both A and R are true, but R is not the correct explanation of A.
Explanation:
Assertion (A) is TRUE because according to the Law of Conservation of Mass, the total mass of reactants equals the total mass of products. Here:
- Mass of hydrogen = 2 g
- Mass of oxygen = 16 g
- Total mass of reactants = 2 + 16 = 18 g
So, 18 g of water is formed.
Reason (R) is also TRUE because Dalton’s Atomic Theory does state that atoms combine in simple whole number ratios to form compounds.
However, R is NOT the correct explanation of A because the assertion is based on the Law of Conservation of Mass (total mass of reactants = total mass of products), not on Dalton’s postulate about whole number ratios. The reason talks about the ratio of combination of atoms, whereas the assertion is about the conservation of total mass during a chemical reaction. These are two different concepts.
Therefore, both statements are independently true, but the reason does not correctly explain the assertion.
Pause and Ponder (Page No. 170)
8. Nitrogen has five valence electrons. Draw the structure of the nitrogen molecule (N₂).
Answer: Nitrogen has atomic number 7 and its electronic configuration is 2, 5. So it has 5 valence electrons in its outermost shell. It needs 3 more electrons to complete its octet.
To achieve stability, two nitrogen atoms each share 3 electrons with each other, forming a triple bond.
Each nitrogen atom has 5 valence electrons and needs 3 more to complete its octet. So two nitrogen atoms share 3 electrons each, forming a triple covalent bond (N≡N).
9. The atomic number of fluorine is 9. Explain the formation of the fluorine molecule (F₂).
Answer: Fluorine has atomic number 9 and its electronic configuration is 2, 7. It has 7 valence electrons in its outermost shell and needs only 1 more electron to complete its octet.
To become stable, two fluorine atoms each share 1 electron with each other, forming a single covalent bond.
Pause and Ponder (Page No. 171)
10. Show the formation of the following molecules:
(i) Carbon dioxide (CO₂)
(ii) Hydrogen sulfide (H₂S)
(iii) Ammonia (NH₃)
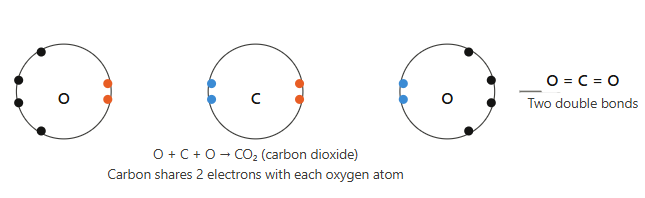
(i) Carbon dioxide (CO₂)
Answer:
Carbon has atomic number 6, electronic configuration 2, 4 → needs 4 electrons to complete octet.
Oxygen has atomic number 8, electronic configuration 2, 6 → needs 2 electrons to complete octet.
So, one carbon atom shares 2 electrons each with two oxygen atoms, forming two double bonds (C=O).
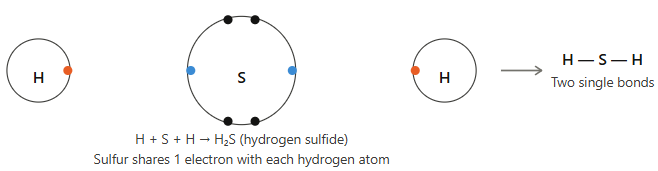
(ii) Hydrogen sulfide (H₂S)
Answer:
Hydrogen has atomic number 1, electronic configuration 1 → needs 1 electron.
Sulfur has atomic number 16, electronic configuration 2, 8, 6 → needs 2 electrons to complete octet.
So, two hydrogen atoms each share 1 electron with sulfur, forming two single bonds (H—S—H).
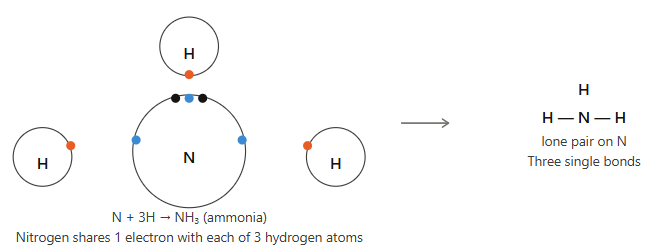
(iii) Ammonia (NH₃)
Answer:
Nitrogen has atomic number 7, electronic configuration 2, 5 → needs 3 electrons to complete octet.
Hydrogen has atomic number 1 → needs 1 electron.
So, three hydrogen atoms each share 1 electron with nitrogen, forming three single bonds.
11. Neon (atomic number 10) neither transfers nor shares its valence electrons. Explain.
Answer: Neon has atomic number 10 and its electronic configuration is 2, 8. This means its outermost shell (L-shell) already has 8 electrons, which is a complete octet.
Since neon’s valence shell is already completely full, it has no need to gain, lose, or share electrons with any other atom. Atoms transfer or share electrons only to achieve a stable octet configuration. Since neon already possesses this stability naturally, it does not participate in any chemical bonding.
This is why neon is called a noble gas — it is chemically inert and exists only as single atoms (monoatomic), not as molecules. Elements like helium, argon, and other noble gases behave similarly because they all have completely filled outer shells.
Pause and Ponder (Page No. 174)
12. What kind of ion will oxygen (O) form?
Answer: Oxygen has atomic number 8 and electronic configuration 2, 6. It has 6 valence electrons and needs 2 more electrons to complete its octet.
Since oxygen gains 2 electrons, it acquires 2 units of negative charge and forms a negatively charged ion called an anion, represented as O²⁻ (oxide ion).
13. Fill in the blanks.
Among magnesium and chlorine, magnesium atom can give two electrons to become Mg²⁺. However, chlorine can take only one electron to become ____________. Now, __________ ion of magnesium and __________ ions of chlorine combine to give magnesium chloride.
Answer:
Among magnesium and chlorine, magnesium atom can give two electrons to become Mg²⁺. However, chlorine can take only one electron to become Cl⁻. Now, one (1) ion of magnesium and two (2) ions of chlorine combine to give magnesium chloride.
Formula: MgCl₂
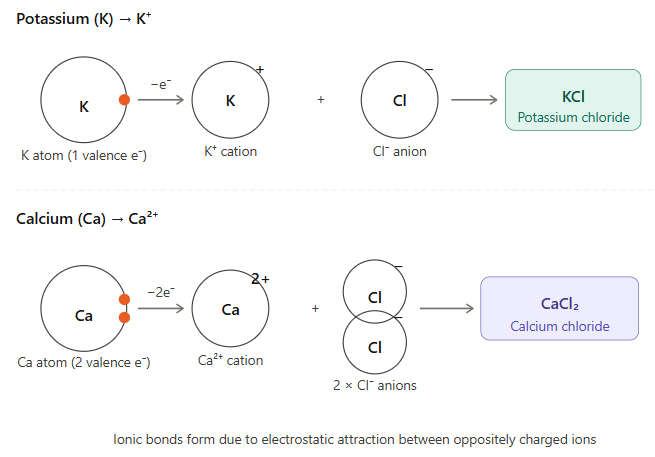
14. Show the formation of cations of potassium (K) and calcium (Ca) atoms, and the formation of their corresponding chlorides using diagrams.
Answer: Formation of cations of K and Ca, and their chlorides:
Explanation:
- Potassium (K): Electronic configuration is 2, 8, 8, 1. It has 1 valence electron. It loses this 1 electron to become K⁺ cation. K⁺ combines with one Cl⁻ to form KCl.
- Calcium (Ca): Electronic configuration is 2, 8, 8, 2. It has 2 valence electrons. It loses both electrons to become Ca²⁺ cation. Ca²⁺ combines with two Cl⁻ ions to form CaCl₂.
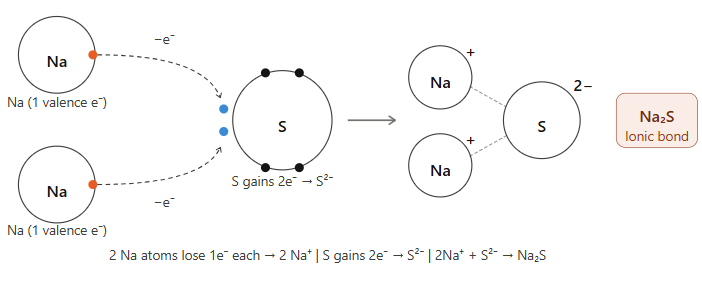
15. Illustrate how sodium sulfide (Na₂S) is formed.
Answer:
Explanation:
- Sodium (Na): Electronic configuration is 2, 8, 1. It has 1 valence electron. Each sodium atom loses 1 electron to form Na⁺ cation.
- Sulfur (S): Electronic configuration is 2, 8, 6. It needs 2 electrons to complete its octet. It gains 2 electrons (one from each sodium atom) to form S²⁻ anion.
- The two Na⁺ ions and one S²⁻ ion are held together by electrostatic force of attraction (ionic bond) to form Na₂S (sodium sulfide).
Reaction: 2Na + S → Na₂S
Pause and Ponder (Page No. 177)
16. Name the following:
(i) CO₂ _________
(ii) NO₂_________
(iii) SF₆ _________
(iv) PCl₃ ________
(i) CO₂
Carbon has 1 atom (mono- is dropped for first element) and oxygen has 2 atoms (di-).
Answer: Carbon dioxide
(ii) NO₂
Nitrogen has 1 atom and oxygen has 2 atoms (di-).
Answer: Nitrogen dioxide
(iii) SF₆
Sulfur has 1 atom and fluorine has 6 atoms (hexa-).
Answer: Sulfur hexafluoride
(iv) PCl₃
Phosphorus has 1 atom and chlorine has 3 atoms (tri-).
Answer: Phosphorus trichloride
17. Write the formula for the following:
(i) Sodium hydrogencarbonate ___________
(ii) Sulfur dioxide ___________________
(iii) Ferric chloride __________________
(iv) Cuprous oxide ________________
(i) Sodium hydrogencarbonate
Na⁺ (valency 1) and HCO₃⁻ (valency 1) → criss-cross gives:
Answer: NaHCO₃
(ii) Sulfur dioxide
Sulfur (1 atom) + oxygen with prefix di- (2 atoms):
Answer: SO₂
(iii) Ferric chloride
Ferric = Fe³⁺ (valency 3), Chloride = Cl⁻ (valency 1) → criss-cross gives:
Answer: FeCl₃
(iv) Cuprous oxide
Cuprous = Cu⁺ (valency 1), Oxide = O²⁻ (valency 2) → criss-cross gives:
Answer: Cu₂O
18. Write the formulae for the compounds formed from the following pairs of ions:
(i) Fe³⁺ and OH⁻
(ii) K⁺ and CO₃²⁻
Answer:
Explanation:
(i) Fe³⁺ and OH⁻
- Fe has charge 3+ and OH has charge 1−
- Criss-cross: Fe gets subscript 1 (omitted), OH gets subscript 3
- Since OH is a polyatomic ion with subscript 3, brackets are used
- Formula: Fe(OH)₃ — Iron (III) hydroxide
(ii) K⁺ and CO₃²⁻
- K has charge 1+ and CO₃ has charge 2−
- Criss-cross: K gets subscript 2, CO₃ gets subscript 1 (omitted)
- Formula: K₂CO₃ — Potassium carbonate
Pause and Ponder (Page No. 179)
19. What type of chemical bond is present in a solid compound that does not conduct electricity in the solid state but conducts electricity when dissolved in water?
Answer: The type of chemical bond present in such a compound is an Ionic Bond.
Explanation: Ionic compounds do not conduct electricity in the solid state because their ions are held in fixed positions by strong electrostatic forces and cannot move freely. However, when dissolved in water, the ions become free to move, allowing the compound to conduct electricity. This behaviour is characteristic of ionic compounds like sodium chloride (NaCl) and copper sulfate (CuSO₄).
20. Metal M, with two electrons in its valence shell (M shell), reacts with oxygen to form a compound that is slightly soluble in water. Predict its:
(i) formula
(ii) type of bond
(iii) electrical conductivity of its aqueous solution.
Answer: Metal M has two electrons in its valence shell (M shell), which means it belongs to the third period and has the electronic configuration 2, 8, 2 — this is Magnesium (Mg).
(i) Formula:
Magnesium has a valency of 2 (forms Mg²⁺), and oxygen has a valency of 2 (forms O²⁻). Using the criss-cross method, since both valencies are equal, the formula simplifies to:
MgO (Magnesium Oxide)
(ii) Type of Bond:
Since magnesium is a metal and oxygen is a non-metal, magnesium transfers its 2 valence electrons to oxygen. This electron transfer forms an Ionic Bond.
(iii) Electrical Conductivity of its Aqueous Solution:
The aqueous solution of MgO will conduct electricity, because when MgO dissolves in water, it dissociates into Mg²⁺ and O²⁻ ions (or forms Mg²⁺ and OH⁻ ions), which are free to move and carry electric charge.
21. Find the molecular mass of nitric acid HNO₃).
Atomic mass — H = 1 u; N = 14 u; O = 16 u.
Answer: Atomic masses:
H = 1 u, N = 14 u, O = 16 u
63 u
22. Find the molecular mass of methane (CH₄).
Atomic mass — C = 12 u; H = 1 u.
Answer: Atomic masses:
C = 12 u, H = 1 u
16 u
Pause and Ponder (Page No. 180)
23. Find the formula unit mass of potassium chloride (KCl).
Atomic mass— K = 39 u; Cl = 35.5 u.
Answer: Atomic masses:
K = 39 u, Cl = 35.5 u
74.5 u
24. Find the formula unit mass of magnesium hydroxide, Mg(OH)₂.
Atomic mass — Mg = 24 u; O = 16 u; H = 1 u.
Answer: Atomic masses:
Mg = 24 u, O = 16 u, H = 1 u
58 u



Leave a Reply