Revise, Reflect, Refine
1. Choose the correct options and explain the reason for the correct and incorrect options in the context of Ernest Rutherford’s gold foil experiment:
(i) The experiment clearly showed the existence of neutrons in the nucleus.
(ii) The results disproved the plum pudding model and led to the idea of a nucleus at the centre of the atom.
(iii) The large deflection of a few alpha particles indicated that most of the mass of the atom and positive charge are packed into a tiny centre.
(iv) The way alpha particles were deflected showed that electrons move around the nucleus.
(i) The experiment clearly showed the existence of neutrons in the nucleus.
Answer: Incorrect
- Neutrons were not discovered in this experiment
- They were discovered later by James Chadwick
So, this statement is wrong.
(ii) The results disproved the plum pudding model and led to the idea of a nucleus at the centre of the atom.
Answer: Correct
- The experiment showed that positive charge is not spread out
- It is concentrated in a small region (nucleus)
So, Thomson’s model was rejected.
(iii) The large deflection of a few alpha particles indicated that most of the mass of the atom and positive charge are packed into a tiny centre.
Answer: Correct
- A few particles were deflected strongly or bounced back
- This proved that the centre is very dense and positively charged
(iv) The way alpha particles were deflected showed that electrons move around the nucleus.
Answer: Incorrect
- The experiment did not give information about electron movement
- It only explained the nucleus and empty space in atom
Correct options: (ii) and (iii)
Incorrect options: (i) and (iv)
2. Which of the following statements are correct or incorrect according to the Bohr’s atomic model? Give a reason for each statement.
(i) Electrons lose energy while moving in fixed orbits and slowly fall into the nucleus.
Answer: Incorrect
- According to Bohr, electrons in fixed orbits do not lose energy
- Therefore, they do not fall into the nucleus
(ii) Electrons can exist anywhere around the nucleus with no fixed energy.
Answer: Incorrect
- Bohr said electrons can exist only in fixed energy levels (orbits)
- They cannot exist anywhere freely
(iii) Electrons revolve around the nucleus in orbits of fixed energy without losing energy.
Answer: Correct
- This is the main idea of Bohr’s model
- Electrons move in fixed orbits with constant energy
(iv) Electrons can be found between energy levels as they move around the nucleus.
Answer: Incorrect
- Electrons cannot exist between energy levels
- They can only jump from one level to another by gaining or losing energy
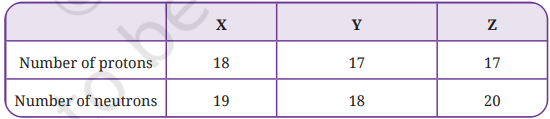
3. The composition of the nuclei of three atomic species X, Y, and Z are given as follows.
Explain the relation between the following:
(i) Y and Z
(ii) Z and X
Answer: First, let us find the Atomic Number and Mass Number of X, Y and Z:
| X | Y | Z | |
|---|---|---|---|
| Number of protons | 18 | 17 | 17 |
| Number of neutrons | 19 | 18 | 20 |
| Atomic number | 18 | 17 | 17 |
| Mass number | 37 | 35 | 37 |
- X → Atomic number = 18, Mass number = 37 (Argon)
- Y → Atomic number = 17, Mass number = 35 (Chlorine)
- Z → Atomic number = 17, Mass number = 37 (Chlorine)
(i) Relation between Y and Z:
Y and Z are ISOTOPES of each other.
- Both Y and Z have the same atomic number = 17 but different mass numbers (Y = 35, Z = 37).
- Since atomic number is the same but mass numbers differ, they are isotopes.
- Both belong to the same element Chlorine (Cl) and are written as ³⁵₁₇Cl and ³⁷₁₇Cl.
- Their chemical properties are similar but physical properties differ due to different masses.
(ii) Relation between Z and X:
Z and X are ISOBARS of each other.
- Both Z and X have the same mass number = 37 but different atomic numbers (Z = 17, X = 18).
- Since mass numbers are same but atomic numbers differ, they are isobars.
- Z is Chlorine (Cl) and X is Argon (Ar) — they are different elements written as ³⁷₁₇Cl and ³⁷₁₈Ar.
- Isobars have different chemical properties because they have different numbers of electrons.
4. What conclusion did Rutherford draw about the position and characteristics of the atom’s positively charged part based on the few alpha particles that bounced back or were deflected at large angles in the gold foil experiment?
Answer: Based on the back-scattering and large angle deflection of a few alpha particles, Rutherford drew the following conclusions:
- The positive charge is not spread throughout the atom but is concentrated in a very small, dense region at the centre called the nucleus.
- The nucleus is extremely dense and heavy, containing most of the mass of the atom, which is why it could deflect heavy alpha particles so sharply.
- The nucleus is extremely small — about 10⁵ times smaller than the atom, with diameter approximately 10⁻¹⁵ m.
- Since most alpha particles passed straight through, most of the atom is empty space.
5. Explain and arrange the following statements in the correct chronological order to show how atomic models have evolved over time.
(i) Bohr’s model proposed that electrons move in fixed orbits around the nucleus, each with a definite energy.
(ii) Thomson’s model depicted the atom as a ʻplum puddingʼ with electrons embedded in a sphere of positive charge.
(iii) Rutherford’s model proposed that atoms have a dense central nucleus.
(iv) Dalton’s model described atoms as indivisible particles.
Answer: (iv) Dalton → (ii) Thomson → (iii) Rutherford → (i) Bohr
- 1st — Dalton’s Model (1808):
Dalton proposed that atoms are indivisible, solid particles and the fundamental building blocks of matter. - 2nd — Thomson’s Model (1897):
Thomson proposed the plum pudding model, where electrons are embedded in a uniform sphere of positive charge, like plums in a pudding. - 3rd — Rutherford’s Model (1911):
Rutherford proposed the nuclear model, stating that atoms have a dense positively charged nucleus at the centre with electrons revolving around it in empty space. - 4th — Bohr’s Model (1913):
Bohr proposed that electrons revolve in fixed orbits called energy levels or shells, each with a definite energy, without losing energy.
6. Electrons move around the nucleus in orbits. Why do they not fly away from the atom? Explain what keeps them attracted to the nucleus.
Answer: Electrons do not fly away from the atom due to the following reasons:
- Electrons carry negative charge and protons in the nucleus carry positive charge. The electrostatic force of attraction between them continuously pulls electrons towards the nucleus, preventing them from flying away.
- As electrons revolve in circular orbits, the outward tendency is perfectly balanced by the inward electrostatic attraction from the nucleus, keeping electrons in stable orbits.
- According to Bohr’s model, electrons revolve in fixed allowed energy levels called stationary states, where they do not lose energy. This keeps them stable in their respective shells without flying outward or falling inward.
7. Assertion(A):The discovery of subatomic particles helped in understanding the atomic structure.
Reason (R): The number of electrons is equal to the number of protons in an atom.
Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Answer: (ii) Both A and R are true, but R is not the correct explanation of A.
Assertion (A) is TRUE:
The discovery of subatomic particles greatly helped in understanding atomic structure. For example:
- Discovery of electrons by Thomson led to the plum pudding model.
- Discovery of the nucleus by Rutherford led to the planetary model.
- Discovery of neutrons by Chadwick explained why atoms are heavier than just their protons.
Each discovery gave scientists a better understanding of how atoms are structured. So Assertion A is correct.
Reason (R) is also TRUE:
In a neutral atom, the number of electrons is always equal to the number of protons, because the total negative charge of electrons must balance the total positive charge of protons. For example, Carbon has 6 protons and 6 electrons. So Reason R is also correct.
However, R is NOT the correct explanation of A:
The fact that electrons equal protons explains the electrical neutrality of an atom, but it does not explain how the discovery of subatomic particles helped in understanding atomic structure. These are two completely separate facts. The Assertion talks about the role of subatomic particle discoveries in building atomic models, while the Reason talks about charge balance. Therefore R does not logically explain A.
8. Magnesium is essential for many biological processes, including muscle contraction. For an atom of magnesium with a mass number of 24 and atomic number 12, determine the number of
(i) protons,
(ii) neutrons,
(iii) electrons, and also illustrate the arrangement of electrons in a magnesium atom.
Answer:
Given:
- Element = Magnesium (Mg)
- Mass number (A) = 24
- Atomic number (Z) = 12
(i) Number of Protons:
Number of protons = Atomic number = 12
(ii) Number of Neutrons:
- Number of neutrons = Mass number − Atomic number
- Number of neutrons = 24 − 12 = 12
(iii) Number of Electrons:
- Since the atom is electrically neutral, number of electrons = number of protons = 12
Arrangement of Electrons (Electronic Configuration):
Using the formula 2n² for maximum electrons in each shell:
- K shell (n=1) → maximum 2 electrons → 2 electrons
- L shell (n=2) → maximum 8 electrons → 8 electrons
- M shell (n=3) → remaining electrons → 12 − 2 − 8 = 2 electrons
Electronic Configuration of Magnesium = 2, 8, 2
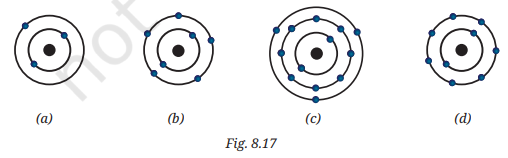
9. Find the following information for the elements shown in Fig. 8.17:
(i) Name of the element
(ii) Symbol
(iii) Total number of electrons
(iv) Number of valence electrons
(v) Valency of the element
(vi) Number of protons
(vii) Atomic number
Answer:Reading the diagrams:
- (a) → 1 shell, 1 electron → K=1
- (b) → 2 shells, 2+1=3 electrons → K=2, L=1
- (c) → 3 shells, 2+8+7=17 electrons → K=2, L=8, M=7
- (d) → 2 shells, 2+1… let’s count → K=2, L=6 = 8 electrons → K=2, L=6
Element (a):
| Information | Answer |
|---|---|
| (i) Name | Hydrogen |
| (ii) Symbol | H |
| (iii) Total electrons | 1 |
| (iv) Valence electrons | 1 |
| (v) Valency | 1 |
| (vi) Number of protons | 1 |
| (vii) Atomic number | 1 |
Element (b):
| Information | Answer |
|---|---|
| (i) Name | Lithium |
| (ii) Symbol | Li |
| (iii) Total electrons | 3 |
| (iv) Valence electrons | 1 |
| (v) Valency | 1 |
| (vi) Number of protons | 3 |
| (vii) Atomic number | 3 |
Element (c):
| Information | Answer |
|---|---|
| (i) Name | Chlorine |
| (ii) Symbol | Cl |
| (iii) Total electrons | 17 |
| (iv) Valence electrons | 7 |
| (v) Valency | 1 (gains 1 electron to complete octet) |
| (vi) Number of protons | 17 |
| (vii) Atomic number | 17 |
Element (d):
| Information | Answer |
|---|---|
| (i) Name | Oxygen |
| (ii) Symbol | O |
| (iii) Total electrons | 8 |
| (iv) Valence electrons | 6 |
| (v) Valency | 2 (gains 2 electrons to complete octet) |
| (vi) Number of protons | 8 |
| (vii) Atomic number | 8 |
Summary Table:
| (a) | (b) | (c) | (d) | |
|---|---|---|---|---|
| Name | Hydrogen | Lithium | Chlorine | Oxygen |
| Symbol | H | Li | Cl | O |
| Total electrons | 1 | 3 | 17 | 8 |
| Valence electrons | 1 | 1 | 7 | 6 |
| Valency | 1 | 1 | 1 | 2 |
| Protons | 1 | 3 | 17 | 8 |
| Atomic number | 1 | 3 | 17 | 8 |
10. Both Rutherford’s and Bohr’s models have electrons orbiting the nucleus. Why did Rutherford’s model fail to explain atomic stability, while Bohr’s model succeeded?
Answer: Rutherford’s Model — Failed to explain stability:
According to Rutherford, electrons revolve around the nucleus in circular orbits. But as learned in physics, a particle moving in a circular path is constantly changing direction, which means it is always accelerating. According to classical physics, any accelerating charged particle must continuously lose energy by emitting radiation. As the electron loses energy, it should spiral inward and eventually fall into the nucleus. This would mean atoms should collapse within a fraction of a second — but in reality atoms are perfectly stable. So Rutherford’s model could not explain atomic stability.
Bohr’s Model — Successfully explained stability:
Bohr solved this problem by introducing the concept of stationary states or fixed energy levels (K, L, M, N shells). He proposed that:
- Electrons can only revolve in certain fixed allowed orbits.
- While moving in these fixed orbits, electrons do not lose any energy.
- The energy of an electron in a stationary state remains constant.
This concept successfully explained why electrons neither lose energy nor fall into the nucleus, making atoms stable.
11. An atom ⁷⁰X has 31 electrons. How many neutrons are there in its nucleus?
Answer:
Given:
- Mass number (A) = 70
- Number of electrons = 31
- Therefore, Number of protons = 31 (atom is neutral)
- Atomic number = 31
Solution:
- Mass number = Protons + Neutrons
- 70 = 31 + Neutrons
- Neutrons = 70 − 31 = 39
Number of neutrons = 39
(This element is Gallium — Ga)
12. An atom has 79 protons and a mass number of 197. Calculate (i) the number of neutrons, and (ii) the number of electrons.
Answer:
Given:
- Number of protons = 79
- Mass number (A) = 197
- Atomic number = 79
(i) Number of Neutrons:
- Mass number = Protons + Neutrons
- 197 = 79 + Neutrons
- Neutrons = 197 − 79 = 118
Number of neutrons = 118
(ii) Number of Electrons:
- Since the atom is electrically neutral, Number of electrons = Number of protons = 79
Number of electrons = 79
(This element is Gold — Au)
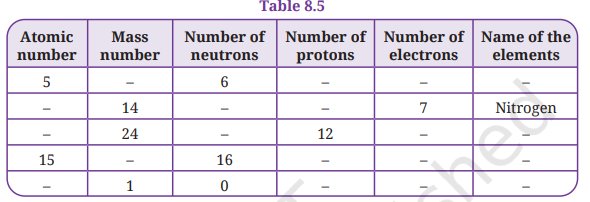
13. Complete the Table 8.5:
Answer:
Row 1: Atomic number = 5, Neutrons = 6
- Number of protons = Atomic number = 5
- Number of electrons = 5 (neutral atom)
- Mass number = Protons + Neutrons = 5 + 6 = 11
- Name of element = Boron (B)
Row 2: Mass number = 14, Electrons = 7, Name = Nitrogen
- Number of protons = Electrons = 7
- Atomic number = 7
- Number of neutrons = Mass number − Protons = 14 − 7 = 7
- Name = Nitrogen (N)
Row 3: Mass number = 24, Protons = 12
- Atomic number = 12
- Number of electrons = 12 (neutral atom)
- Number of neutrons = Mass number − Protons = 24 − 12 = 12
- Name of element = Magnesium (Mg)
Row 4: Atomic number = 15, Neutrons = 16
- Number of protons = Atomic number = 15
- Number of electrons = 15 (neutral atom)
- Mass number = Protons + Neutrons = 15 + 16 = 31
- Name of element = Phosphorus (P)
Row 5: Mass number = 1, Neutrons = 0
- Number of protons = Mass number − Neutrons = 1 − 0 = 1
- Atomic number = 1
- Number of electrons = 1 (neutral atom)
- Name of element = Hydrogen (H)
Completed Table 8.5:
| Atomic number | Mass number | Neutrons | Protons | Electrons | Name |
|---|---|---|---|---|---|
| 5 | 11 | 6 | 5 | 5 | Boron (B) |
| 7 | 14 | 7 | 7 | 7 | Nitrogen (N) |
| 12 | 24 | 12 | 12 | 12 | Magnesium (Mg) |
| 15 | 31 | 16 | 15 | 15 | Phosphorus (P) |
| 1 | 1 | 0 | 1 | 1 | Hydrogen (H) |
14. Aman was discussing the structure of atom with his classmates. During the discussion, he learnt that an element X has a mass number of 35 and contains 18 neutrons. Based on this information, answer the following questions:
(i) How many electrons and protons does element X have?
(ii) What is its atomic number?
(iii) Identify the element X.
(iv) Write its electronic configuration.
(v) How many valence electrons does it have?
(vi) What will be the mass number if two neutrons are added to its nucleus?
(vii) What will be the relation of X with the new atom?
Answer:
Given:
- Mass number (A) = 35
- Number of neutrons = 18
(i) Number of electrons and protons:
- Number of protons = Mass number − Neutrons
- Number of protons = 35 − 18 = 17
- Since atom is electrically neutral, Number of electrons = 17
Protons = 17, Electrons = 17
(ii) Atomic number:
- Atomic number = Number of protons = 17
Atomic number = 17
(iii) Identify element X:
- Atomic number 17 corresponds to Chlorine (Cl)
Element X is Chlorine (Cl)
(iv) Electronic configuration:
- Total electrons = 17
- K shell = 2
- L shell = 8
- M shell = 17 − 2 − 8 = 7
Electronic configuration = 2, 8, 7
(v) Number of valence electrons:
- Valence shell is M shell
- Valence electrons = 7
(vi) Mass number after adding two neutrons:
- New number of neutrons = 18 + 2 = 20
- New mass number = Protons + Neutrons = 17 + 20 = 37
New mass number = 37
(vii) Relation of X with the new atom:
- Original atom X = ³⁵₁₇Cl (protons = 17, neutrons = 18)
- New atom = ³⁷₁₇Cl (protons = 17, neutrons = 20)
- Both have the same atomic number (17) but different mass numbers (35 and 37)
X and the new atom are ISOTOPES of each other.
They are both Chlorine atoms but with different numbers of neutrons. Their chemical properties are similar because they have the same electronic configuration, but their physical properties differ due to different masses.
15. In an atom, there are 12 protons and 12 neutrons in the nucleus. Now, imagine that all the electrons are replaced with some hypothetical particles that have the same charge as electrons but are 500 times heavier. What effect will this replacement have on the atom’s:
(i) Atomic number
(ii) Atomic mass
(iii) Mass number
(iv) Overall charge
Answer:
Given:
- Number of protons = 12
- Number of neutrons = 12
- Atomic number = 12 → Element is Magnesium (Mg)
- Electrons are replaced with hypothetical particles having same charge as electrons but 500 times heavier
(i) Effect on Atomic Number:
- Atomic number depends only on the number of protons in the nucleus.
- The replacement affects only electrons, not protons.
- Number of protons remains 12.
Atomic number remains UNCHANGED = 12
(ii) Effect on Atomic Mass:
- Original electrons have negligible mass.
- The hypothetical particles are 500 times heavier than electrons.
- Mass of one electron = 9.1 × 10⁻³¹ kg
- Mass of hypothetical particle = 500 × 9.1 × 10⁻³¹ kg
- There are 12 such hypothetical particles replacing 12 electrons.
- This adds a significant amount of mass to the atom.
Atomic mass will INCREASE significantly because the hypothetical particles are 500 times heavier than electrons, contributing considerable mass unlike normal electrons whose mass is negligible.
(iii) Effect on Mass Number:
- Mass number = Number of protons + Number of neutrons
- Mass number = 12 + 12 = 24
- Mass number is calculated based only on protons and neutrons in the nucleus.
- The replacement of electrons does not affect protons or neutrons.
Mass number remains UNCHANGED = 24
(Note: Mass number is a count of nucleons only, not actual mass. So even though atomic mass increases, mass number stays the same.)
(iv) Effect on Overall Charge:
- The hypothetical particles have the same charge as electrons (i.e., −1 each).
- The number of these particles = 12 (same as original electrons).
- Total negative charge = 12 × (−1) = −12
- Total positive charge from protons = 12 × (+1) = +12
- Total charge = +12 + (−12) = 0
Overall charge remains UNCHANGED = 0 (neutral atom)
Since the hypothetical particles carry the same charge as electrons, the atom remains electrically neutral.
Summary Table:
| Property | Before Replacement | After Replacement |
|---|---|---|
| Atomic number | 12 | 12 (unchanged) |
| Atomic mass | ~24 u | Increases significantly |
| Mass number | 24 | 24 (unchanged) |
| Overall charge | 0 (neutral) | 0 (neutral, unchanged) |

Leave a Reply