Notes For All Chapters Biology Class 11 CBSE
All living organisms require a continuous supply of energy for their survival. Energy is used to carry out various functions such as uptake of materials, absorption, growth, development, movement and even breathing. About 50% of the energy produced by the cell is utilized by these cellular activities and rest of it is changed into heat and get lost. Now, the question arises from where does this energy comes to carry out all these processes of life.
Topic 1 Respiration : The Basics
We eat food in order to obtain energy. The food we eat in the form of macro molecules is oxidized to fulfill energy requirement of body for caring out all the basic life processes.
As we have already studied in last chapter, that only green plants and cyanobacteria can prepare their own food by the process called photosynthesis. They use trapped energy in order to obtain their food by converting light energy into chemical energy, which thereby gets stored into the bonds of carbohydrates such as glucose, sucrose, starch, etc.
But all cells, tissues and organs in plants do not photosynthesise instead, photosynthesis takes place in only some parts of plants, i.e., only cells that contain chloroplasts (mosdy areas located in superficial layers).
Hence, all other organs, tissues and cells in green plant that are non-green are need food for oxidation. Hence, food has to be translocated from the green parts to the non-green parts for oxidation processes.
Need of Photosynthesis
Animals on the other hand are heterotrophic in nature, i.e., they either obtain their food directly from the plants (herbivores) or indirectly, dependent on herbivores for their food (carnivores).
Saprophytes are dependent on dead and decaying matter for their food (e.g., Fungi). Thus, it can be concluded that all the food that is respired for life processes ultimately comes from photosynthesis.
Cellular Respiration
Cellular respiration or the mechanism of breakdown of food materials within-the cell to release energy and trapping the same energy for synthesis of ATP.
Respiration is the process of breaking of the C-C bonds of complex compounds through oxidation within the cells, leading to release of considerable amount of energy.
It is to be noted that site of breaking down of complex molecules to yield energy is cytoplasm and mitochondria (also only in eukaryotes) which is different from the site of photosynthesis, which is chloroplast in plants.
Respiratory Substrates
The compounds that are oxidised during the process of respiration are called respiratory substrates. Carbohydrates are used as major respiratory substrates are oxidised in high amounts, to release energy, but under some conditions in some plants, proteins, fats and organic acids are also used as respiratory substrates.
Differences between Respiration and Combustion
ATP : Energy Currency of the Cell
During the process of oxidation of food within a cell, all the energy contained in the respiratory substrates is not released free into the cell, or in a single step. Instead it gets released in a series of step-wise reactions controlled by enzymes and is trapped as chemical energy in the form of ATP.
Hence, the energy released in respiration by the process of oxidation is not used directly but is used in synthesising ATP (which is utilised whenever energy needs to be utilised).
Thus, it is said that ATP acts as the energy currency of the cell. The energy trapped in ATP is utilised in many energy requiring processes of the organisms, and the carbon skeleton produced during respiration is used as precursors for the biosynthesis of other molecules in a cell.
Do Plants Breathe : Exchange of Gases in Plants
For the process of respiration, plant takes O2 and releases CO2. Plants have stomata and lenticles for gaseous exchange instead of specialised organs that are present in animals for exchange of gases.
Following are the reasons which shows, how plants can get along without respiratory organs
(i) Every part of the plant has the ability to take care of its own needs of gas exchange and also very little transport of gases occur from one part of the plant to another.
(ii) It is only during the process of photosynthesis that large volumes of gases are exchanged and each leaf of the plant has ability to take care of its own needs during these periods.
Thus, when cells photosynthesis, availability of O2 is not a problem in these cells due to a continuous release of O2 that takes place within the cell.
(iii) Gases may easily diffuse in large, bulky plants as distance for diffusion is not so great because living cells in a plant are located quite close to the surface of the plant.
In case of stems, which are thick and woody in nature, the organisation of living cells is in the form of thin layers, which are found inside and beneath the bark. Like leaves which have stomata for gaseous exchange, these stems also have openings called lenticels. Internal cells are dead and provide only mechanical support to the plant.
This depicts that most cells of plant have atleast a part of their surface in contact with the air. The loose packing of parenchyma cells in leaves, stems and roots and provides an interconnected network of air spaces helps in facilitating this process.
Types of Respiration
We know that during the process of respiration, utilisation of O2 takes place with the release of CO2, water and energy as products.
According to the dependence of cells on oxygen, cellular respiration may be classified into two types as given below
1 Aerobic Respiration
This is the type of respiration in which organism utilise oxygen for the complete oxidation of organic food into CO2 and water. It occurs inside the mitochondria.
Aerobic respiration yields more energy as the respiratory substrate gets completely oxidised in the presence of O2.
2. Anaerobic Respiration
This is the type of respiration in which organic food is oxidised incompletely without utilising energy as oxidant. It occurs in cytoplasm and often releases small amount of energy.
It is believed that the first cells on this planet lived in an oxygen free environment, i.e., they were anaerobes. Even among present day living organisms, several are adapted to anaerobic conditions.
Some of them are facultative anaerobes (organisms that have capability of switching from aerobic to anaerobic conditions according to the availability of oxygen) while others are obligate anaerobes (organisms that are killed by normal atmospheric concentration of oxygen of 21%).
Thus, in any case, all living organisms retain the enzymatic machinery for partial oxidation of glucose in the absence of oxygen. And this breakdown of glucose to pyruvic acid is called glycolysis.
Topic 2 Respiration: The Mechanism
Cellular respiration occurs inside the cell and proceeds with the help of enzymes. The first step in respiration (taking glucose as substrate) is the glycolysis (glucose oxidised to pyruvic acid). After which the pyruvic acid may enter the Krebs’ cycle (aerobic respiration) or undergo fermentation (anaerobic respiration).
Glycolysis
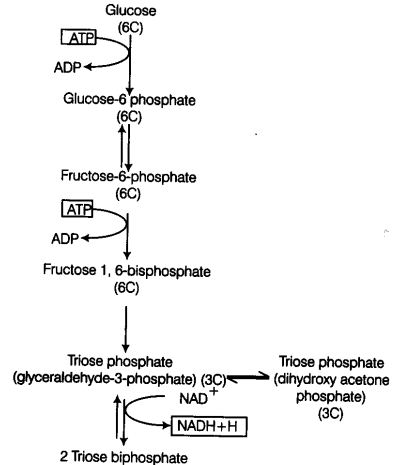
Glycolysis (Gr. Glycor-sugar; lysis-splitting), is a step-wise process by which one molecule of glucose (6C) breaks down into two molecules of pyruvic acid (3C).
The scheme of glycolysis was given by Gustav Embden, Otto Meyerhof and J Parnas and is often referred as the EMP pathway. It is a common pathway in both aerobic and anaerobic modes of respiration. But in case of anaerobic organisms, it is the only process of respiration.
Glycolysis occurs in the cytoplasm of the cell. During the process glucose gets partially oxidised. In plants this glucose is derived from sucrose (end product of photosynthesis) or from storage carbohydrates.
During the course of process in plant this sucrose is first converted into glucose and fructose by the action of invertase enzyme after this, these two monosaccharides enter the glycolytic pathway.
Steps Involved in Glycolysis
In glycolysis, a chain of 10 reactions often reactions occur under the control of different enzymes.
It involves the following steps
Step I Phosphorylation of glucose occur under the action of an enzyme hexokinase and Mg2+ that gives rise to glucose-6-phosphate by the utilisation of ATP.
Step II Isomerisation of this phosphorylated glucose-6-phsophate takes place to form fructose-6-phosphate with the help of an enzyme phosphohexose isomerase (Reversible Reaction).
Step III This fructose-6-phosphate is again phosphorylated by ATP in order to form fructose 1, 6-bisphosphate in the presence of an enzyme phosphofructokinase and Mg2+.
The steps of phosphorylation of glucose to fructose 1, 6-bisphosphate (i.e., from step 1 to 3) activates the sugar thus, preventing it from getting out of the cell.
Step IV Splitting of fructose 1, 6-bisphosphate takes place into two triose phosphate molecules, i.e., dihydroxyacetone 3-phosphate and 3-phosphoglyceraldehyde (i.e., PGAL). This reaction is catalysed by an enzyme aldolase.
Step V Each molecule of PGAL removes two redox equivalents in the form of hydrogen atom and transfer them to a molecule of NAD+ (This NAD+ forms NADH + H+) and accepts inorganic phosphate (Pi) from phosphoric acid. This reaction in turn leads to the conversion to PGAL (which gets oxidised) to 1, 3-bisphosphoglycerate (BPGA) (Reversible reaction).
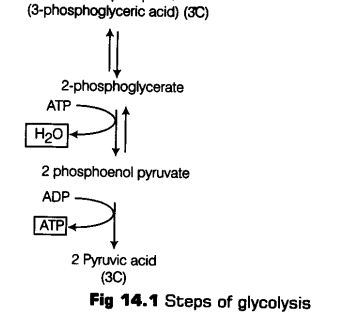
Step VI 1, 3-bisphosphoglycerate is converted to 3-phosphoglycerate with the formation of ATP.
This reaction is catalysed by an enzyme phosphoglycerate kinase.
It is also known as energy yielding process. The formatibn of ATP directly from metabolites constitutes substrate level phosphorylation (Reversible reaction).
Step VII In the next step, 3-phosphoglycerate is subsequently isomerised to form 2-phosphoglycerate, catalysed by enzyme phosphoglyceromutase (Reversible reaction).
Step VIII In the presence of enzyme enolase and Mg2+, with the loss of a water molecule, 2-phosphoglycerate is converted to Phosphoenol Pyruvate (PEP) (Reversible reaction).
Step IX High energy phosphate group of Phosphoenol Pyruvate (PEP) is transferred to a molecule of ADP, by the action of enzyme pyruvate kinase in the presence of Mg2+ and K+ This in turn produces two molecules of pyruvic acid (pyruvate) and a molecule of ATP by substrate level phosphorylation. The pyruvic acid thus, produced is the key product of glycolysis.
Metabolic Fate of Glycolysis
The overall reaction of glycolysis can be depicted as
* Glucose + 2Pi + 2ADP + 2NAD+ —> 2 Pyruvate + 2ATP + 2NADH + 2H+
* Two molecules of NADH on oxidation produce 6 molecules of ATP. Therefore, a net gain of 8ATP molecules occurs during glycolysis.
* The fete of glycolysis depends upon the availability of oxygen in the cell. In the presence of oxygen, pyruvic acid will enter the mitochondrion and undergo complete oxidation of glucose to CO 2 andH20 in aerobic respiration (Krebs’ cycle).
* On the other hand in the absence of oxygen, the pyruvic acid will undergo anaerobic respiration (lactic acid fermentation or alcoholic fermentation).
Note:
* Kostytcher (1902) coined the term anaerobic respiration.
* Glycolysis has two phases, i.e., preparatory (glucose is broken down to glyceraldehyde-3-phosphate) and pay off phase (the GAL-3-PD4 is changed into pyruvate producing NADH and ATP).
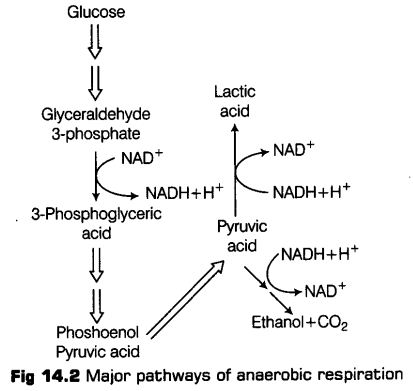
Fermentation
Various microorganisms, bacteria, animals and plants are known to catabolise pyruvic acid into various organic compounds depending upon the specific enzymes they possess.
Some of these types are as follows
(i) During alcoholic fermentation, in fungi (e.g., yeast), and some higher plants, the incomplete oxidation of glucose is achieved under anaerobic condition by a series of reactions in which pyruvic acid is converted to CO2 and ethanol.
It is done under two steps
(а) Pyruvic acid is first decarboxylated to acetaldehyde in the presence of enzyme pyruvic acid decarboxylase.
(b) This acetaldehyde is further reduced to ethyl alcohol or ethanol in the presence of enzyme, i.e., alcohol dehydrogenase.
(ii) During lactic acid fermentation, organisms like some bacteria produces lactic acid as an end product from pyruvic acid.
During the reduction, the pyruvic acid produced in glycolysis is reduced by NADH2 to form lactic acid, CO2 is not produced and NADH2 is oxidised to NAD+.
This reaction is catalysed by an lactic acid dehydrogenase, FMN proteins and Zn2+ ions.
Likewise, in case of animal cells also (such as muscles) during exercise, when there is inadequate amount of oxygen for cellular respiration, pyruvic acid is reduced to lactic acid by lactate dehydrogenase.
Thus, in both the processes reoxidation of reducing (NADH + H+) agent takes place.
Energy Yield in Fermentation
In both alcoholic and lactic acid fermentation, the energy released is very less, i.e., not more than 7% of the energy is released from glucose and not all of it is trapped as high energy bonds of ATP.
Also, the fermentation processes are proved to be hazardous in nature because either acid or alcohol is produced on oxidation. Apart from this, yeasts may also poison themselves to death if the concentration of alcohol reaches about 13%.
Drawback of this process is that organisms cannot carryout complete oxidation of glucose and are also unable to extract out the energy stored to synthesise a larger number of ATP molecules required for cellular metabolism.
Differences between Glycolysis and Fermentation
Aerobic Respiration
Aerobic respiration is the next step (after glycolysis) that leads to complete oxidation of organic substances. It occurs in the presence of oxygen. The oxygen acts as a final acceptor of electron and protons are removed from the substrate. For aerobic respiration to take place within the mitochondria, the final product of glycolysis, i.e., pyruvic acid is transported into from the cytoplasm mitochondria and thus, the second phase of respiration is initiated.
The process of aerobic respiration involves two crucial events
(i) The complete oxidation of pyruvate occurs by the step-wise removal of all the hydrogen atoms, thereby, leaving three molecules of CO2. This occurs in the matrix of mitochondria.
(ii) The electrons removed as part of the hydrogen atoms are then passed on to molecular 02 with the simultaneous synthesis of ATP. This on the contrary takes place on the inner membrane of the mitochondria.
Oxidative Decarboxylation of Pyruvic Acid
In mitochondria, pyruvic acid (formed by the glycolytic catabolism of carbohydrates in cytosol) undergoes oxidative decarboxylation (i.e., removal of CO2 in aerobic conditions) forming a key compound, i.e., acetyl Co-A by the action of pyruvic acid dehydrogenase (in mitochondrial matrix) through a series of reactions.
Thus, acetyl Co-A acts as a connecting link between glycolysis and citric acid cycle.
During this process, two molecules of NADH are produced from the metabolism of two molecules of pyruvic acid (produced from one glucose molecule during glycolysis).
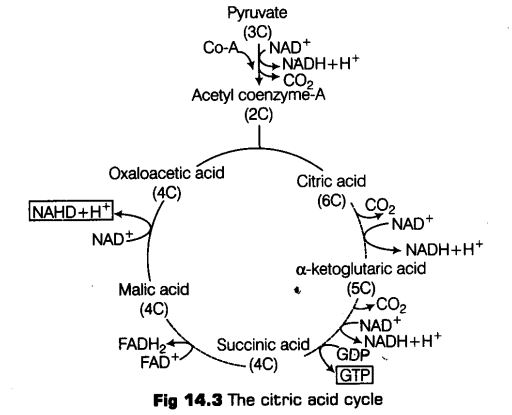
Tricarboxylic Acid (TCA) Cycle
The acetyl Co-A then enters a cyclic pathway, Krebs’ cycle (or tricarboxylic acid cycle, TCA) in mitochondrial matrix. Various coenzymes including NAD+ and Co-A also participates in the reaction catalysed by pyruvic acid dehydrogenase.
It was first elucidated by Sir Hans Kreb, a British Biochemist in 1940.
The whole cycle explains how pyruvate is broken down to CO2 and water.
Following are the steps of Krebs ’ cycle
(i) Condensation The Krebs’ cycle starts with the condensation of acetyl group with oxaloacetic acid and water to yield citric acid, a 6C compound. This is the first stable product of the cycle.
This step is catalysed by an enzyme citrate synthetase. Co-A is liberated during this reaction.
(ii) Citric acid then undergoes reorganisation in two steps in order to form in the presence of an enzyme acinotase. intermediate
(iii) Oxidative decarboxylation Isocitrate is followed by two successive steps of oxidative decarboxylation, that leads to the formation of a-ketoglutaric acid, (a 5C compound in the presence of an enzyme isocitrate dehydrogenase and Mn1 ) and then succinyl Co-A, catalysed by a-complex. .
The succinyl Co-A then splits into a 4C compound succinic acid and Co-A with the addition of water. During this conversion, a molecule of GTP (guanosine triphosphate) is synthesised catalysed by an enzyme succinyl Co-A synthetase (this occurs when co-enzyme A transfers its high energy to a phosphate group that joins GDP forming GTP).
(i) GTP is also an energy carrier like ATP. Thus, this is the only high energy phosphate produced in the Krebs’ cycle.
(ii) In plants cells, this reaction also produces ATP from ADP.
In the remaining steps of Krebs’ cycle, succinyl Co-A is oxidised to oxaloacetic acid, a 4C compound following the formation of fumaric acid and malic acid catalysed by enzymes succinate dehydrogenase and fumacase respectively.
Output of Krebs’ Cycle or Citric Acid Cycle
During this cycle of reactions, 3 molecules of NAD+ are reduced to NADH + H+, and one molecule of FAD+ is reduced to FADH2. And also one molecule of ATP is reduced directly from GTP (by substrate level phosphorylation).
For continuous oxidation of acetyl Co-A, continued replenishment of oxaloacetic acid is necessary. In addition to this regeneration of NAD+ and FAD+ from NADH and FADH2 respectively are also required.
The summary equation for this phase of respiration is as follows
Till now, glucose has been broken down to release CO2 and 8 molecules of NADH+H+, two FADH2 are synthesised and just two molecules of ATP.
Importance of Citric Acid Cycle
The citric acid cycle is important in thefollowing ways
(i) This is the major pathway for the formation for ATP molecules.
(ii) Many intermediate compounds of this cycle are used in the synthesis of other biomolecules.
Differences between glycolysis and Krebs’cycle

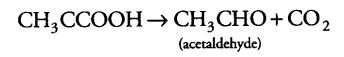
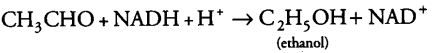

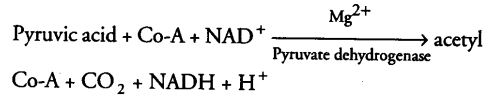
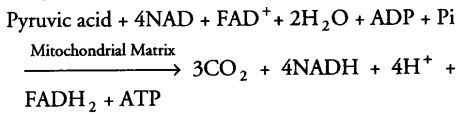


Leave a Reply