Notes For All Chapters Chemistry Class 11 CBSE
• Hydrocarbon
A compound of carbon and hydrogen is known as hydrocarbon.
• Saturated Hydrocarbon
A hydrocarbon is said to be saturated if it contains only C—C single bonds.
For example: Ethane CH3—CH3
• Unsaturated Hydrocarbon
• Aromatic Hydrocarbon
Benzene and its derivatives are called aromatic compounds.
Example:
• Alicyclic Compounds
Cyclic compounds which consist only of carbon atoms are called alicyclic or carboeyclic compounds.
• Heterocyclic Compounds
Cyclic compounds in which the ring atoms are of carbon and some other element (For example, N, S, or O) are called heterocyclic compounds.
• Alkanes
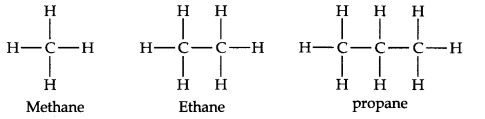
Alkanes are the simplest organic compounds made of carbon and hydrogen only.
They have the general formula CnHC2n+2 (where n = 1, 2, 3, etc.)
The carbon atoms in their molecules are bonded to each other by single covalent bonds. Since the carbon skeleton of alkanes is fully saturated’ with hydrogens, they are also called saturated hydrocarbons. Alkanes contain strong C —C and C —H bonds. Therefore, this class of hydrocarbons are relatively chemically inert. Hence they are sometimes referred to as paraffins (Latin parum affinis = little affinity). First three members of this class can be represented as
Structure:
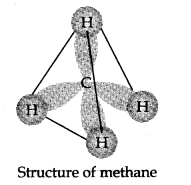
In methane carbon forms single bonds with four hydrogen atoms. All H—G—H bond angles are of 109.5°. Methane has a tetrahedral structure. C—C and C—H bonds are formed by head-on overlapping of sp3 hybrid orbitals of carbon and Is orbitals of hydrogen atoms.
• Nomenclature Guidelines
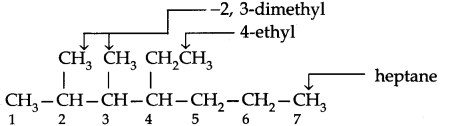
Use the following step-by-step procedure to write the IUPAC names from the structural formulas. Consider the following structural formula:
Step 1. Identify the longest chain: In the given example, longest chain has seven carbons. The seven carbon chain is heptane.
Step 2. Number the chain: The chain is numbered from left to right. This gives lowest numbers to the attached alkyl group.
Step 3. Identify the alkyl group: There are two methyl groups at C-2 and C-3, there is one ethyl group of C-4.
Step 4. Write the IUPAC name: In this case the IUPAC name is 4-Ethyl-2,3-dimethyl heptane. Always keep in mind (a) Numbers are separated from each other by commas. (b) Numbers are separated from names by hyphens, (c) Prefixes di, tri are not taken into account in alphabetising substituent names.
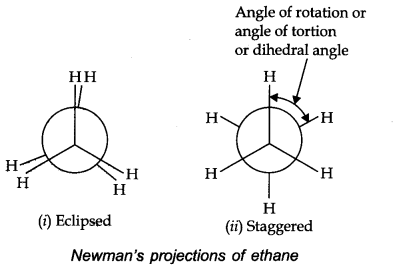
• Newman Projections
In this projection, the molecule is viewed at the C—C bond head on.
• Relative Stability of Conformations
In staggered form of ethane there are maximum repulsive forces, minimum energy and maximum stability of molecule. On the other hand, when the staggered form changes in the eclipsed form the electron clouds of the carbon hydrogen bonds come closer to each other resulting in increase in electron cloud repulsions, molecule have to possess more energy and thus has lower stability.
Torsional Angle: Magnitude of torsional strain depends upon the angle of rotation about C—C bond. This angle is also called dihedral angle or torsional angle.
• Alkenes
Alkenes are hydrocarbons that contain a carbon-carbon double bond (C=C) in their molecule.
They have the general formula
Structure:
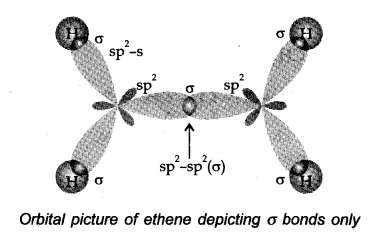
Let us consider (H2C=CH2) for illustrating the orbital make up of alkenes.
In ethylene the carbon atoms are sp2 hybridized- They are attached to each other by a a bond and a σ bond.
The a bond results from the overlap of two sp2 hybrid orbitals. The π bond is formed from overlap of the unhybridized p-orbitals. Ethylene is a planar molecule.
Ppints to be noted
(i) The carbon-carbon double bond in alkenes is made up of one σ and one π-bond.
(ii) Alkenes are more reactive than Alkanes. This is due to the availability of n electrons.
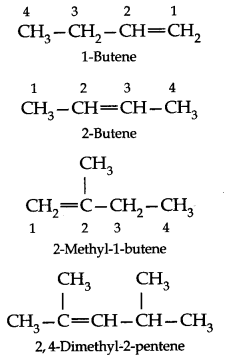
• Nomenclature
In IUPAC system
(i) The name of the hydrocarbon is based on the parent alkene having the longest ‘ carbon chain of which double bond is apart.
(ii) This chain is numbered from the end near the double bond and its position is indicated by the number of the carbon atom not which the double bond originates,
(iii) The name of the parent alkene with the position number of the double bond is written first and then the names of other substituents prefixed to it.
(iv) When there are two or three double bonds in a molecule, the ending-one of the corresponding alkane is replaced by-a diene to get the name.
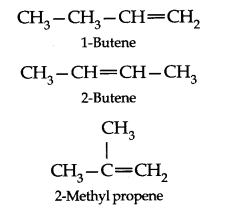
• Isomerism
Structural Isomerism: Ethene and propene have no structural isomers, but there are three structures of butenes.
Of these, two are straight chain structures with the difference being in the position of double bond in the molecules.
These are position isomers and third structure is a branched-chain isomer.
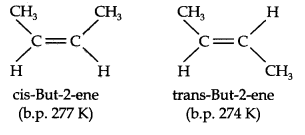
Geometrical Isomerism: It is known that a carbon-carbon double bond is made up of one σ bond and one π-bond. The π-bond presents free rotation about the double bond.
This presentation of rotation about the carbon-carbon double bond gives rise to the phenomenon of geometrical isomerism. An alkene having a formula RCH=CHR can have two stereoisomers, depending upon whether the two alkyl groups are on the same or opposite sides of the double bond. If they are on the same side, then it is called cis-isomer. If they are on opposite sides, then it is called trans-isomer.
Due to different arrangement of atoms or groups in space, these isomers differ in their properties like melting point, boiling point, dipole moment, solubility, etc.
• Alkynes
Alkynes are characterised by the presence of a triple bond in the molecule.
Their general formula is CnH2n-2.
The first and the most important member of this series of hydrocarbons is acetylene, HC=CH, and hence they are also called the Acetylenes.
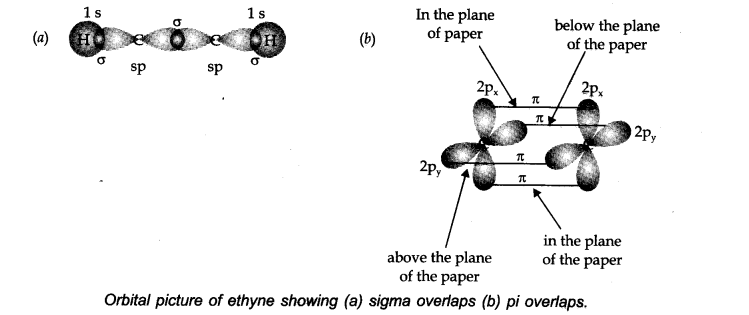
Structure: Let us consider ethyne (HC=CH) for illustrating the orbital make up of ethyne. In ethyne, the carbon atoms are sp hybridized. They are attached to each other by a σ-bond and two π-bonds.
The σ -bond results from the overlap of two sp hybrid orbitals. The π bonds are formed from the separate overlap of the two p-orbitals from the two adjacent carbon atoms.
The other sp hybrid orbital of each carbon atom forms a σ bond with another carbon or hydrogen atom. Ethyne is a linear molecule.
Points to be noted:
(i) The carbon-carbon triple bond in alkynes is made up of one σ and two π bonds.
(ii) Like alkenes, alkynes undergo addition reaction. These reactions are due to the availability of more exposed π electrons.
• Nomenclature
IUPAC System: The IUPAC names of alkynes are obtained by dropping the ending-ane of the parent alkane and adding the suffix-yne. Carbon chain including the triple bond is – numbered from the end nearest this bond. The position of the triple bond is indicated by prefixing the number of carbon preceding it to the name of the alkyne.
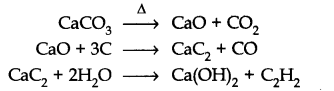
Preparation:
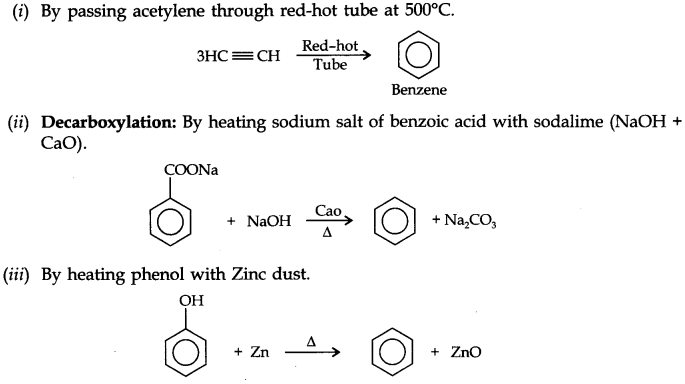
From calcium carbide: Ethyne is prepared by treating calcium carbide with water. Calcium carbide is prepared as follows:
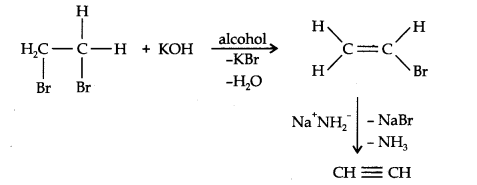
From vicinal dihalides: When reacted with vicinal dihalides, alcoholic potassium hydroxide undergo dehydrohalogenation. One molecule of hydrogen halide is eliminated to form alkenyl halide which on treatment with sodamide gives alkyne.
• Aromatic Hydrocarbons
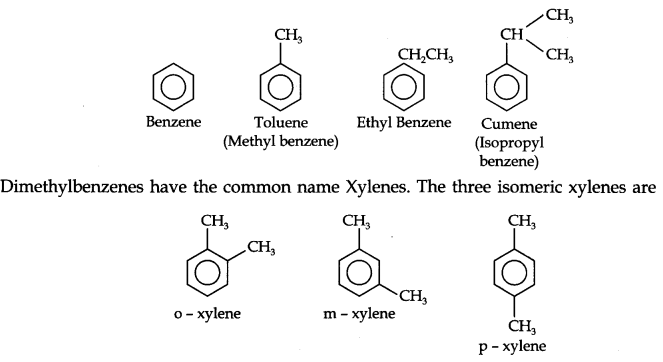
These hydrocarbons are also known as ‘arenes’. Most of such compounds were found to contain benzene ring.
Aromatic compounds containing benzene ring are known as benzenoids and those not containing a benzene ring are known as non-benzenoids. Some examples of arenes are given below.
Nomenclature and Isomerism: Benzene and its homologous are generally called by their common names which are accepted by the IUPAC system. The homologous of benzene having a single alkyl group are named as Alkyl benzenes.
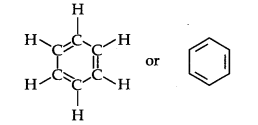
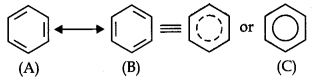
Structure of Benzene: By elemental analysis, it is found that molecular formula of benzene is C6H6. This indicates that benzene is a highly unsaturated compound. In 1865, Kekule gave the cyclic planar structure of benzene with six carbons with alternate double and single bonds.
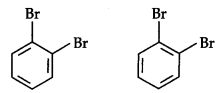
The Kekule structure indicates the possibility of two isomeric 1,2-dibromobenzenes. In one of the isomers, the bromine atoms are attached to the doubly bonded carbon atoms whereas in the other they are attached to the singly bonded carbon.
In fact, only one ortho-dibromobenzene could be prepared.
To overcome this problem Kekule suggested that benzene was a mixture of two forms.
Failure of Kekule’s structure: Kekule structure of benzene failed to explain the unique stability and its preference to substitution reaction than addition reactions.
Resonance Structure of Benzene: The phenomenon in which two or more structures can be written for a substance which involve identical positions of atoms is called resonance. In benzene’s Kekule’s structures (1) and (2) represent the resonance structures. Actual structure – of the molecule is represented by hybrid of the these two structures.
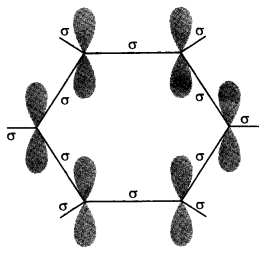
Orbital structure of benzene: All six carbon atoms in benzene are sp2 hybridized. The sp2 hybrid orbitals overlap with each other and with s orbitals of the six hydrogen atoms forming C—C and C—H σ-bonds.
X-Ray diffraction data indicates that benzene is a planar molecule. The data indicates that all the six C—C bond length are of the same order (139 pm) which is intermediate between (C—C) single bond (154 pm) and C—C double bond (133 pm). Thus the presence of pure double bond in benzene gives the idea of reductance of benzene to show addition reaction under normal condition. The is, It explains the unusual behaviour of benzene.
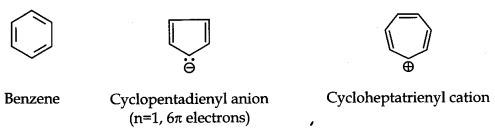
Aromaticity: It is a property of the sp2 hybridized planar rings in which the p orbitals allow cyclic delocalization of π electrons.
Conditions for Aromaticity:
(i) An aromatic compound is cyclic and planar.
(ii) Each atom in an aromatic ring has a p orbital. These p orbitals must be parallel so that a continuous overlap is possible around the ring.
(iii) The cyclic π molecular orbital (electron cloud) formed by overlap of p orbitals must contain (4n + 2) π electrons. Where n = integer (0, 1, 2, 3, etc.). This is known as Huckel rule.
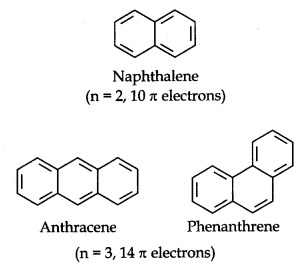
Some Examples of Atomic Compounds are given below:
Preparation of Benzene: Benzene is commercially isolated from coaltar. However, there are some synthetic methods which is applied in the laboratory for the preparation of benzene.
Physical Properties of Benzene:
(i) Benzene is a colourless liquid.
(ii) It is’ insoluble in water. It is soluble in alcohol, ether, chloroform etc.
(iii) Benzene itself is a good solvent for many organic and inorganic substances e.g., fat, resins, sulphur and iodine.
(iv) It bums with a luminous, sooty flame in contrast to alkanes and alkenes which usually bum with a bluish flame.
Chemical Properties:
Benzene undergeos following types of chemical reactions.
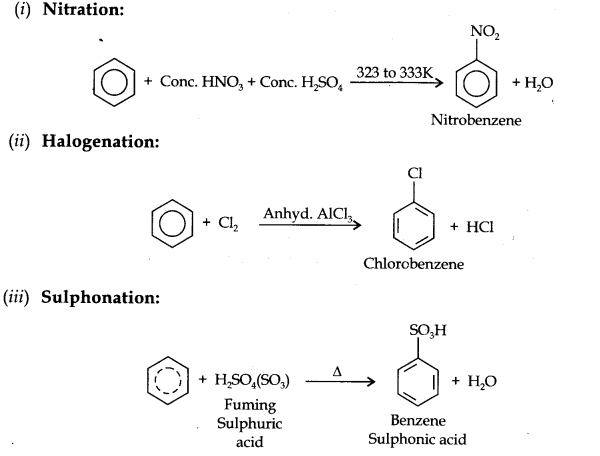
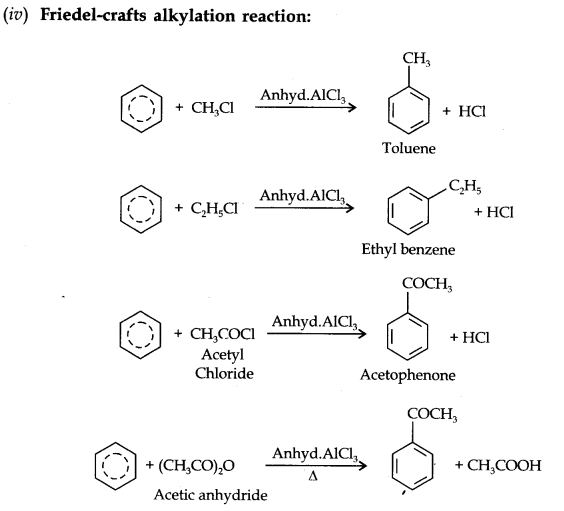
(i) Electrophillic Substitution Reaction
(ii) Addition Reaction
Electrophillic Substitution Reactions:
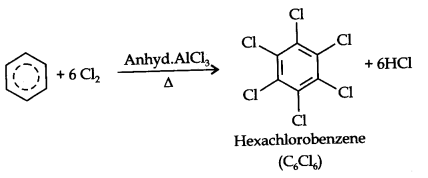
Benzene on treatment with excess of chlorine in the presence of anhydrous AlCl3 can be chlorinated to hexachlorobenzene (C6Cl6)
Mechanism of electrophilic substitution reactions:
All electrophilic substitution reactions follow the same three step mechanism.
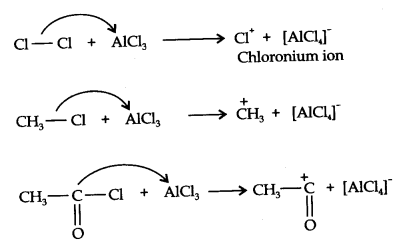
Setp 1. Formation of an electrophile:
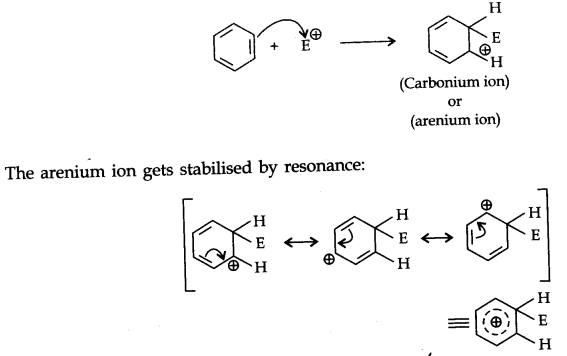
Step 2. The electrophile attacks the aromatic ring to form a carbonium ion.
Step 3. Loss of proton gives the substitution product.
Activating groups: These group activates the benzene ring for the attack by an electrophile. Example, —OH; —NH2, —NHR, —NHCOCH3, —OCH3 —CH3 —C2H5 etc.
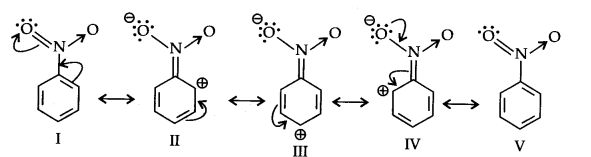
Deactivating groups: Due to deactivating group because of strong —I effect, overall electron density on benzene ring decreases. It makes further substitution difficult.
Metadirecting group: The groups which direct the incoming group to meta position are called meta directing groups. Some examples of meta directing groups are —N02, —CN, —CHO, —COR, —COOH, —COOR, -S03H etc.
Let us consider the example of nitro group: Since Nitro group due to its strong -I effect reduces the electron density in benzene ring. Nitrobenzene is a resonance hybrid of following structures.
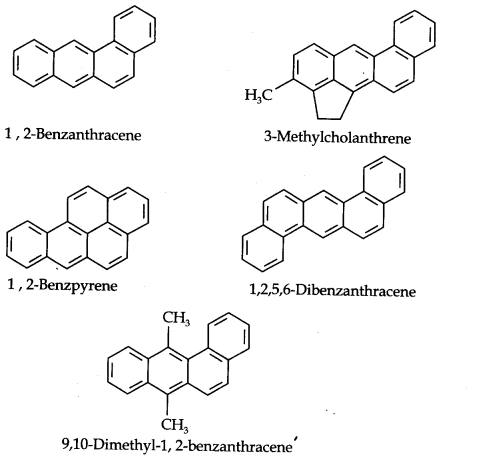
Carcinogenicity and Toxicity: Some polynuclear hydrocarbons containing more than two benzene rings fused together become toxic and they are having cancer producing property. They are actually formed due to incomplete combustion of some organic materials like tobacco, coal and petroleum, etc.
Some of the carcinogenic hydrocarbons are given below.
• Hydrocarbons: They are compounds of carbon and hydrogen only.
Open Chain saturated compound—Alkane
Unsaturated Compound—Alkenes and Alkynes Aromatic Compound—Benzene and its derivatives Terminal alkynes are weakly acidic in nature.
• Conformation: Spatial arrangements obtained by rotation around sigma bonds.
• Eclipsed Conformation: Less stable because of more repulsion between bond pairs of electrons.
• Staggered: It is more stable since there is less repulsion between bond pairs of electrons.
• Geometrical isomerism: Observed only in compounds containing a double bond.
• Stability of benzene. Is explained on the basis of resonance hybrid.
• Arenes: Take part in electrophilic substitution reaction.
Aromaticity is determined by Huckle’s rule (4n + 2) rule
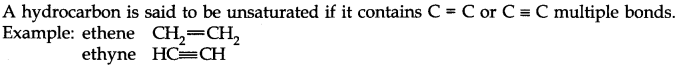
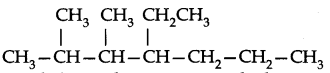
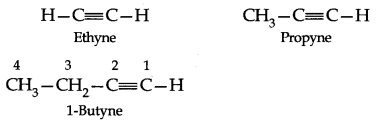

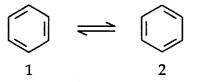

Leave a Reply